What Are Covalent Compounds Composed Of
All the matter in the universe is composed of the atoms of more than 100 different chemical elements which are found both in pure form and combined in chemical compoundsA sample of any given pure element is composed only of the atoms characteristic of that element and. The nucleotide adenosine triphosphate ATP is composed of a ribose sugar an adenine base and three phosphate groups Figure 230.

Properties Ionic Covalent Compounds 1 Covalent Bonding Ionic Intermolecular Force
A mixture is composed of different types of atoms or molecules that are not chemically bonded.

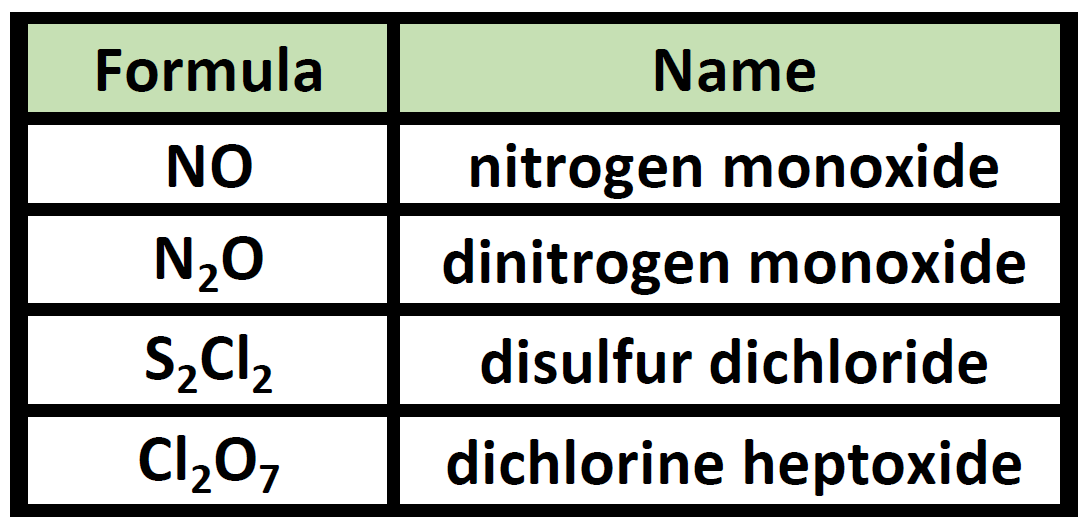
What are covalent compounds composed of. 44 Shape of Covalent Compounds. Chemical compound any substance composed of identical molecules consisting of atoms of two or more chemical elements. A molecule is composed of one or more chemical bonds between molecular orbitals of different atoms.
Covalent organic frameworks COFs 242526 are a class of organic materials that combine crystallinity modular synthetic versatility highly. Covalent bonding and covalent compounds will be. Unlike ionic compounds with their extended crystal lattices covalent molecules are discrete units with specific three-dimensional shapes.
Carbon compounds are defined as chemical substances containing carbon. Organic carbon compounds are far more numerous than inorganic carbon compoundsIn general bonds of carbon with other elements are covalent bondsCarbon is tetravalent but carbon free radicals and carbenes occur as short-lived. A heterogeneous mixture is a mixture of two or more chemical substances where the various components can be visually distinguished.
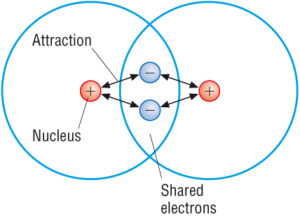
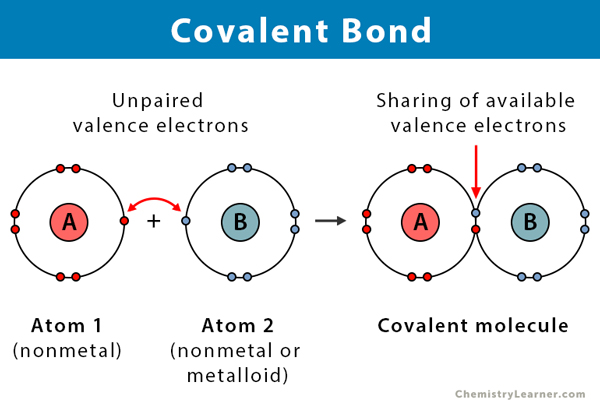
More compounds of carbon exist than any other chemical element except for hydrogen. The second way for an atom to obtain an octet of electrons is by sharing electrons with another atom. These shared electrons simultaneously occupy the outermost shell of both atoms.
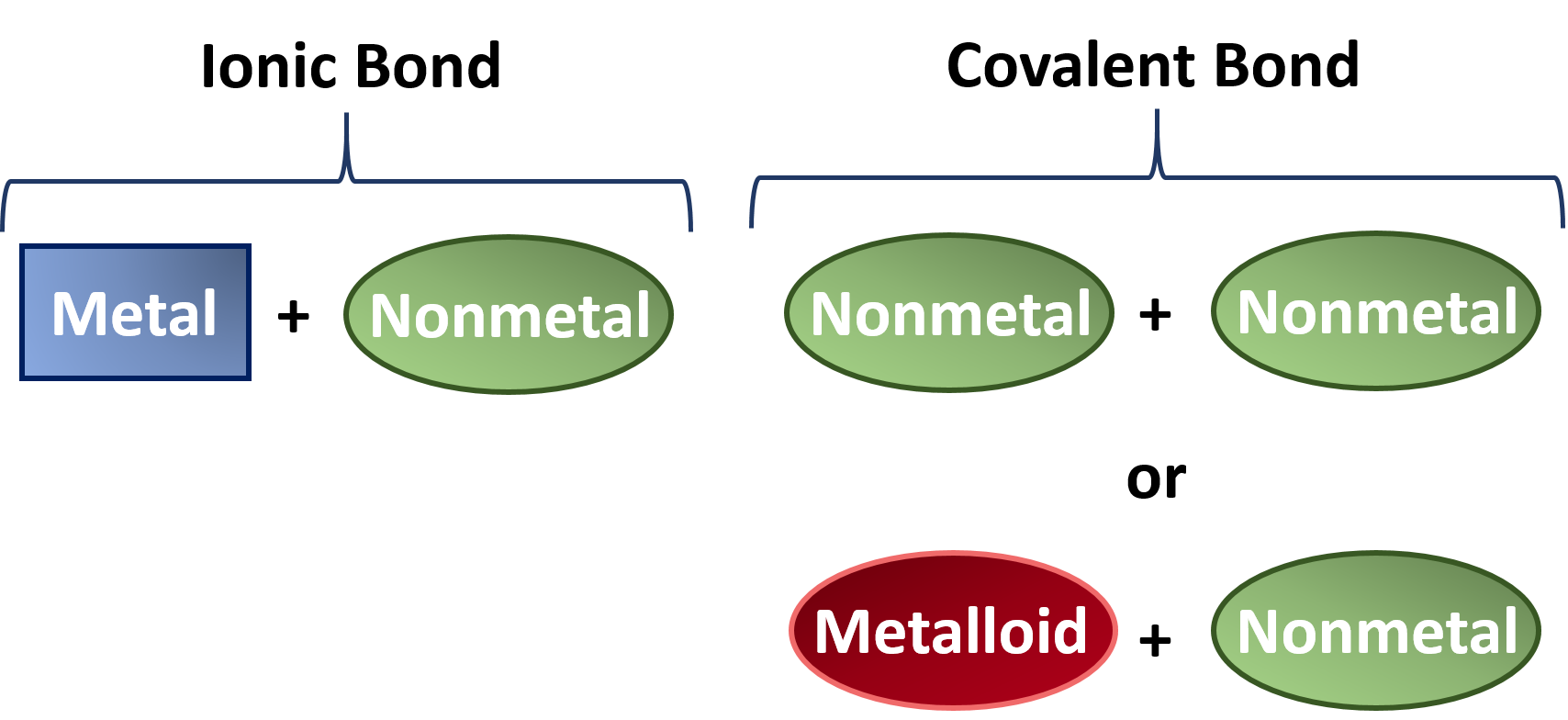
Molecular compounds held together by covalent bonds. The resulting compounds are called ionic compounds. The bond made by electron sharing is called a covalent bond.
A molecule may be polar either as a result of polar bonds due to differences in electronegativity as described above or as a result of an asymmetric arrangement of nonpolar covalent bonds and non-bonding pairs of electrons known as a full. The shape of a molecule is determined by the fact that covalent bonds which are composed of shared negatively charged electrons tend to repel one another. ATP is classified as a high energy compound because the two covalent bonds linking its three phosphates store a significant amount of potential energy.

4 3 Structure And Properties Of Covalent Compounds Sl Youtube
Covalent Compounds Covalent Bond Properties Examples With Videos

Single Covalent Bond Covalent Bonding Bond Chemistry

Characteristics Of Covalent Compounds Chemical Bonding 16h Youtube

How To Name Ionic And Covalent Compounds Chemistrynotes Com

Section 8 1 Are Electrons Only Transferred While Studying Ionic And Metallic Bonding Electrons Have Been Mov Covalent Bonding Ionic Bonding Metallic Bonding

Covalent Bonds Biology For Majors I
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry

Chemistry Lab Molecular Models Of Covalent Compounds Polar Or Nonpolar Chemistry Labs Chemistry Chemistry Classroom
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry

Lewis Dot Structures For Covalent Compounds Part 1 Covalent Bonding Chemistry Classroom Chemistry Lessons
Covalent Bond Definition Types And Examples

Properties Ionic Covalent Compounds Experiment 4 Covalent Bonding Ionic Chemical Bond

Covalent Bond Examples Formation Properties What Is A Covalent Bond Video Lesson Transcript Study Com

Covalent Bond Animation Chemistry Covalent Bonding Chemistry Classroom Chemistry Education

Covalent Compounds Covalent Bond Properties Examples With Videos

Molecular Compound Easy Science Molecular Ionic Bonding Easy Science

Ions Present In Compounds Composed Of Elements With Very Low Very High Electroneg Ie Metals Nonmetals This Ionic Bonding Covalent Bonding Chemical Bond
