What Are Covalent Compounds Made Of
Non-volatile b Usually exist as liquids or gases at room temperature. Covalent compounds like sugar and food coloring can dissolve and diffuse but they do not dissociate.

Covalent Bond Examples Formation Properties What Is A Covalent Bond Video Lesson Transcript Study Com
Volatile c Conduct electricity in the molten state or in an aqueous solution but do not conduct electricity in the solid state.

What are covalent compounds made of. The Ionic Compound is formed when there is a big difference in the electronegativity of the atoms where the less electronegative atom. Covalent bonding is an important and extensive concept in chemistry and it will be treated in considerable detail in a later module. Because of this variety in bonding and complexity carbon atoms can form a tremendous variety of compounds.
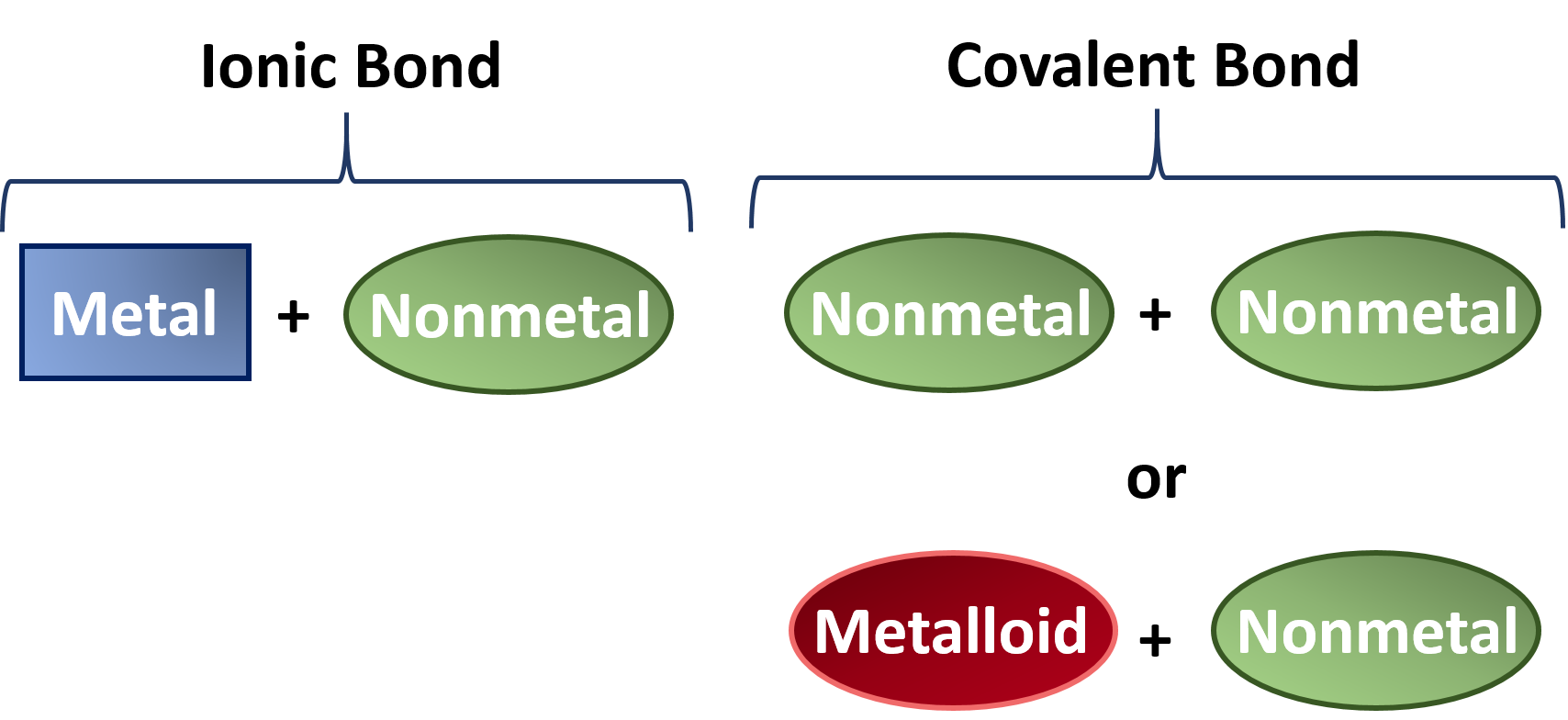
The Strength of Covalent Bonds Depends on the Overlap between the Valence Orbitals of the Bonded Atoms. Element - An element is a substance composed of the same type of atoms eg. Covalent bonding occurs in most non-metal elements and in compounds formed between non-metals.
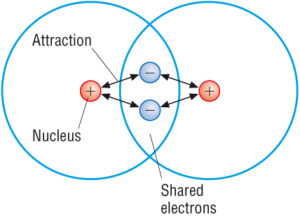
This bond forms between the functional group and a carbon atom from the main body of the molecule. A covalent bond is formed when two atoms share a pair of electrons. Ionic Compounds vs Covalent Compounds.
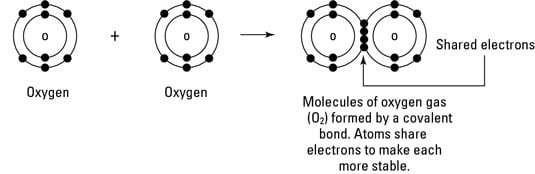
These shared electrons are found. It can be considered as a compound which does not contain carbon to hydrogen bond which calls C-H bond. Many compounds do not contain ions but instead consist solely of discrete neutral molecules.
Compound - A compound is a substance made of more than one type of atom eg. Heavier larger atoms in the same periodic group. Paper chromatography is used to separate mixtures of soluble substances.
Significant sources of VOC are chemical plants gasoline pumps oil-based paints autobody shops and print shops. Difference Between Organic And Inorganic Compounds - Organic compound is a chemical compound of living things which contains Carbon and carbon atoms because of their relations with organisms. Compounds have a constant composition.
Covalent compounds are one of the two categories that compounds are usually grouped into. Without stirring the food coloring will mix into the water through only the movement of the water and food coloring molecules. The law of constant composition states that the ratio by mass of the elements in a chemical compound is always the same regardless of the source of the compound.
Mixtures do notWater is always 888 O and 112 H by. Covalent bonds only form between nonmetallic elements because these elements have the same or similar electronegativity values. Compounds having a particular handedness complex 3D shapes etc.
Electrons move freely and very fast around an atom. Metallic compounds contain freely floating electrons which allow them to conduct electricity and heat well. Water H 2 O carbon dioxide CO 2.
Examples of compounds include table salt or sodium chloride NaCl an ionic compound sucrose a molecule nitrogen gas N 2 a covalent molecule a sample of copper intermetallic and water H 2 O a covalent molecule. Molecule - A molecule is the smallest particle of either an element or a compound. Inorganic compound is a chemical compound which is the opposite of an organic compound.
The different dissolved. Comparison of Properties of Ionic and Covalent Compounds. Inert or Noble Gases.
Here is a list of examples of compounds made of two elements. 234 is a time series of drops of food coloring diffusing in water. For many molecules the sharing of electrons allows each atom to attain the equivalent of a full valence.
Because of the nature of ionic and covalent bonds the materials produced by those bonds tend to have quite different macroscopic properties. Examples of Compounds. Covalent bonds are highly stable bonds with low melting points.
Organic compounds contain what is called functional groups a structural unit made up of groups of atoms in a molecule that are bonded to the rest of the molecule by a covalent bond. 12 Whats So Great About Carbon. Depending on the energy level and closeness to the nucleus the electrons will stay within a certain shape.
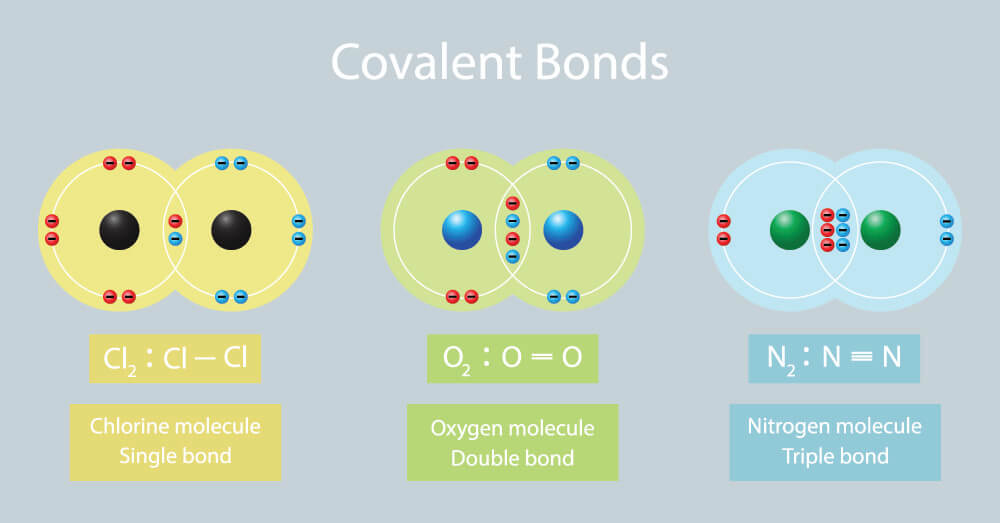
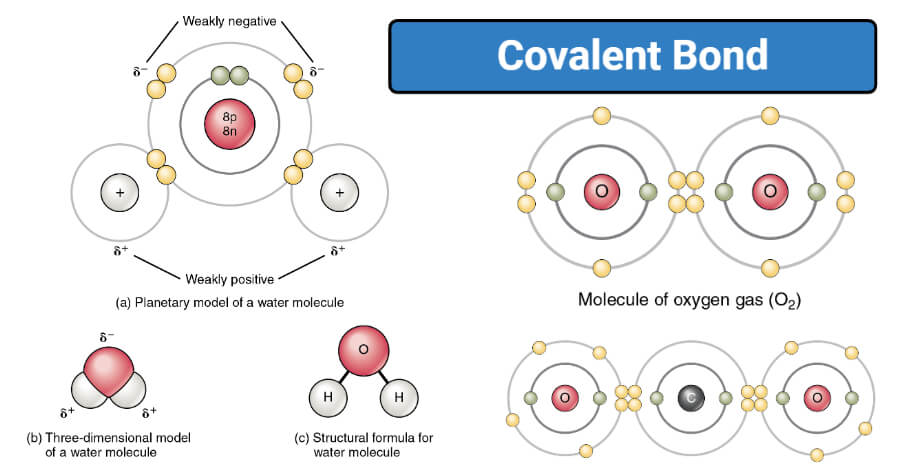
And b two lighter versus two heavier. The relative sizes of the region of space in which electrons are shared between a a hydrogen atom and lighter smaller vs. A covalent bond is a chemical bond that involves the sharing of electron pairs between atomsThese electron pairs are known as shared pairs or bonding pairs and the stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding.
The difference between Ionic Compounds and Covalent Compounds is their formation. Here are examples of covalent compounds and a look at their common properties. The other kind of compound is an ionic compound and ionic compounds are composed of electrically charged molecules or atoms thanks to an electrical imbalance caused by losing or gaining electrons.
A compound is made up of two or more elements chemically bonded together. Molecules made of more than one type of covalently bonded nonmetal atoms like carbon dioxide gas CO2 remain nonpolar if they are symmetrical or if their atoms have relatively equal pull. These are often coloured substances such as food colourings inks dyes or plant pigments.
Ionic compounds tend to be crystalline structures with high melting points that are water soluble. Although some stratospheric ozone is transported into the troposphere and some VOC and NOx occur naturally the majority of ground-level ozone is the result of reactions of man-made VOC and NOx. The atoms of covalent materials are bound tightly to each other in stable molecules but those molecules are generally not very strongly attracted to other molecules in the material.
Covalent compounds or molecular compounds are chemical compounds made of elements connected by covalent bonds. Covalent compounds Ionic compounds composed of simple molecules a Have high melting and boiling points a Have low melting and boiling points b Exist as solids at room temperature. More than 16000000 organic compounds are known as opposed to about 600000 inorganic compounds.
Double covalent bonds are usually made from nonmetals like carbon nitrogen and oxygen. Many covalent compounds are flexible or gaseous and are not water soluble. Gold Au oxygen O 2.
Covalent molecules made of only one type of atom like hydrogen gas H2 are nonpolar because the hydrogen atoms share their electrons equally. The law of constant composition can be used to distinguish between compounds and mixtures of elements. These molecular compounds covalent compounds result when atoms share rather than transfer gain or lose electrons.

Covalent Bond Definition Properties Examples Facts Britannica

Covalent Bond An Overview Sciencedirect Topics
Ch150 Chapter 4 Covalent Bonds And Molecular Compounds Chemistry

Ionic Bond Images Stock Photos Vectors Shutterstock
Covalent Bond Biology Dictionary

Covalent Compounds Covalent Bond Properties Examples With Videos

Covalent Bonds Biology For Majors I
Covalent Bond Definition Properties Types Formation Examples
Covalent Compounds Covalent Bond Properties Examples With Videos
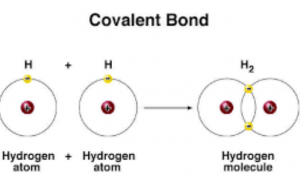
What Are The Types Of Covalent Bonds Science Online

Covalent Bonding Gcse The Science Hive

Covalent Bonding Biology Definition Role Expii
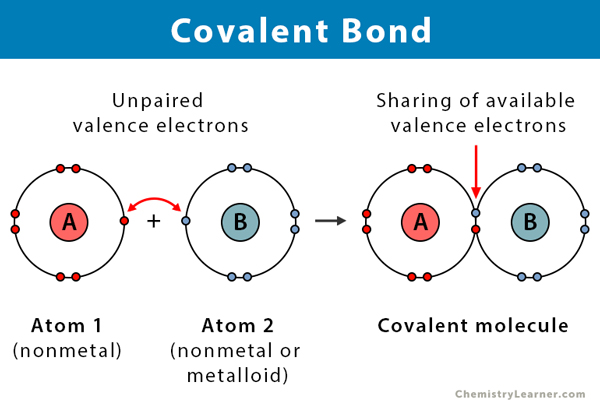
Covalent Bond Definition Types And Examples

Ppt Covalent Compounds Powerpoint Presentation Free Download Id 5168896
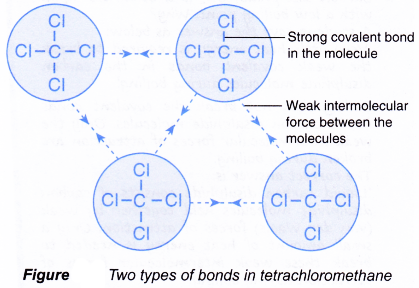
Properties Of Ionic And Covalent Compounds A Plus Topper

Covalent Bond An Overview Sciencedirect Topics
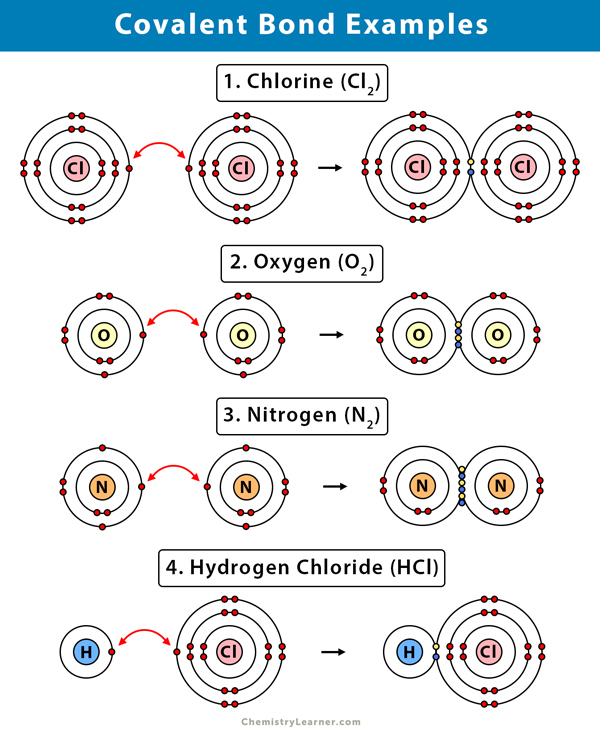
Covalent Bond Definition Types And Examples
Environmental Science What Is Covalent Bonding Dummies
What Does A Covalent Bond Mean Quora