What Are Three Examples Of Inorganic Compound
Inorganic changes are processes like redox reactions oxidation and the mixture of bases and acids. Examples of bases are sodium hydroxide calcium carbonate and potassium oxide.

What Are Inorganic Compounds Definition Characteristics Examples Video Lesson Transcript Study Com
Most bases are minerals that react with acids to form water and salts.

What are three examples of inorganic compound. And Sodium Chloride NaCl. The nomenclature naming systems for IONIC and MOLECULAR compounds are different. The water molecules in inorganic hydrates are only loosely bonded to the compound and there is no chemical reaction involved.
Elements and compounds are the two forms in which pure substances exist. To correctly write the chemical formula for this molecule the two polyatomic ions ammonium and hydroxide are represented separately in the formula. H is the symbol for hydrogen.
The compound ammonium hydroxide has the formula NH 3 OH. Examples of organic chemical changes include halogenation methylation crude oil cracking and polymerization. Compounds Compounds are chemical substances made up of two or more elements that are chemically bound together in a fixed.
A base is a substance that can neutralize the acid by reacting with hydrogen ions. Atom cluster compounds including metal clusters have been unwittingly used by humans since antiquity. Examples of such inorganic compounds include carbon monoxide CO silicon carbide SiC and carbonic acid H 2 CO 3 and salts thereof.
There are a total of three atoms in one molecule of carbon dioxide CO 2. You likely already know some hydrates since some of them are common everyday products. Examples of compounds include water or hydrogen oxide H2O.
If H is the. Most primary and secondary batteries use reactions of inorganic materials such as metals and metal compounds to store energy. The water molecules can be removed from the compound fairly easily such as through heating.
Carbon dioxide CO 2 is another example of an inorganic compound because it does not contain both carbon and hydrogen. While it may seem logical to combine symbols so that the formula reads as NH 4 O this would not be correct. Elements Elements constitute the simplest chemical substances in which all the atoms are exactly the same.
These consist of any positive ion except H combined with any negative ion. One molecule of CO 2 contains one atom of carbon and two atoms of oxygen. There are three types of hydrates.
Here is the list of examples of branches of inorganic compounds found at home. Finally inorganic chemical changes are chemical changes that dont use carbon as a part of the chemical reaction. Examples of elements include oxygen hydrogen sodium chlorine lead iron.
The number 2 as used here means there are 2 atoms of the element hydrogen in every molecule of the compound water. Inorganic organic and gas or clathrate hydrates. All other types of carbon-containing compounds are called organic materials.
Hydrate any compound containing water in the form of H2O molecules usually but not always with a definite content of water by weight. The oldest artificially produced metal cluster may be calomel Hg 2 Cl 2 which was known in India already in the 12th century. The elucidation of the structure of cluster compounds only became possible in the 20th century.
INORGANIC NOMENCLATURE NAMING INORGANIC COMPOUNDS Every compound has its own CHEMICAL FORMULA and its own NAME.
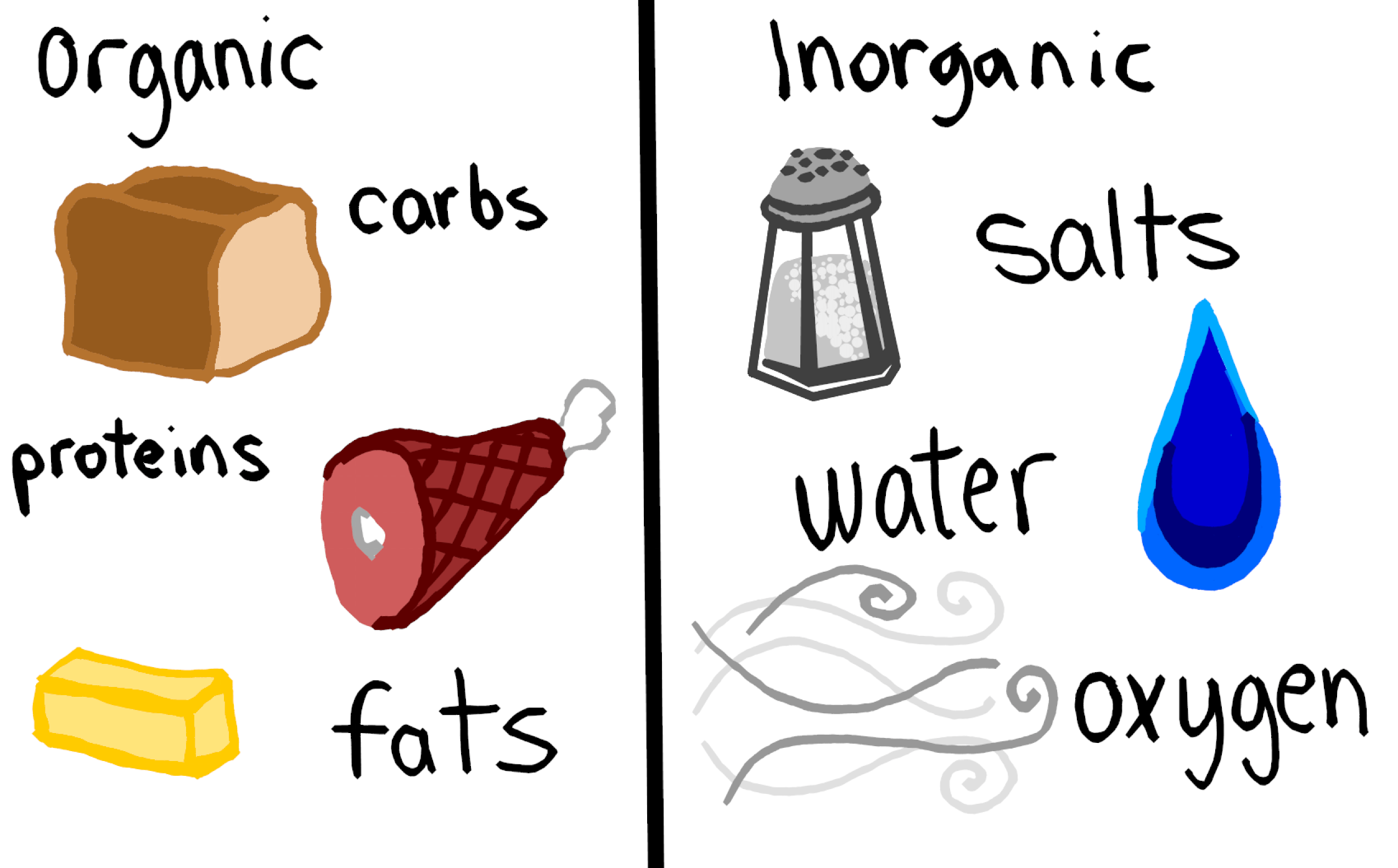
Organic Vs Inorganic Molecules Definition Overview Expii

Inorganic Compounds Anatomy And Physiology

Difference Between Organic And Inorganic Compounds Youtube

Organic Vs Inorganic Molecules Definition Overview Expii

Examples Of Metals And Inorganic Compounds Used In Medicine Rat Oral Download Scientific Diagram
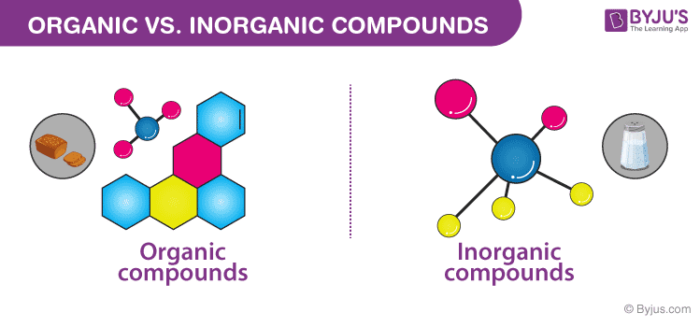
Difference Between Organic And Inorganic Compounds Key Differences
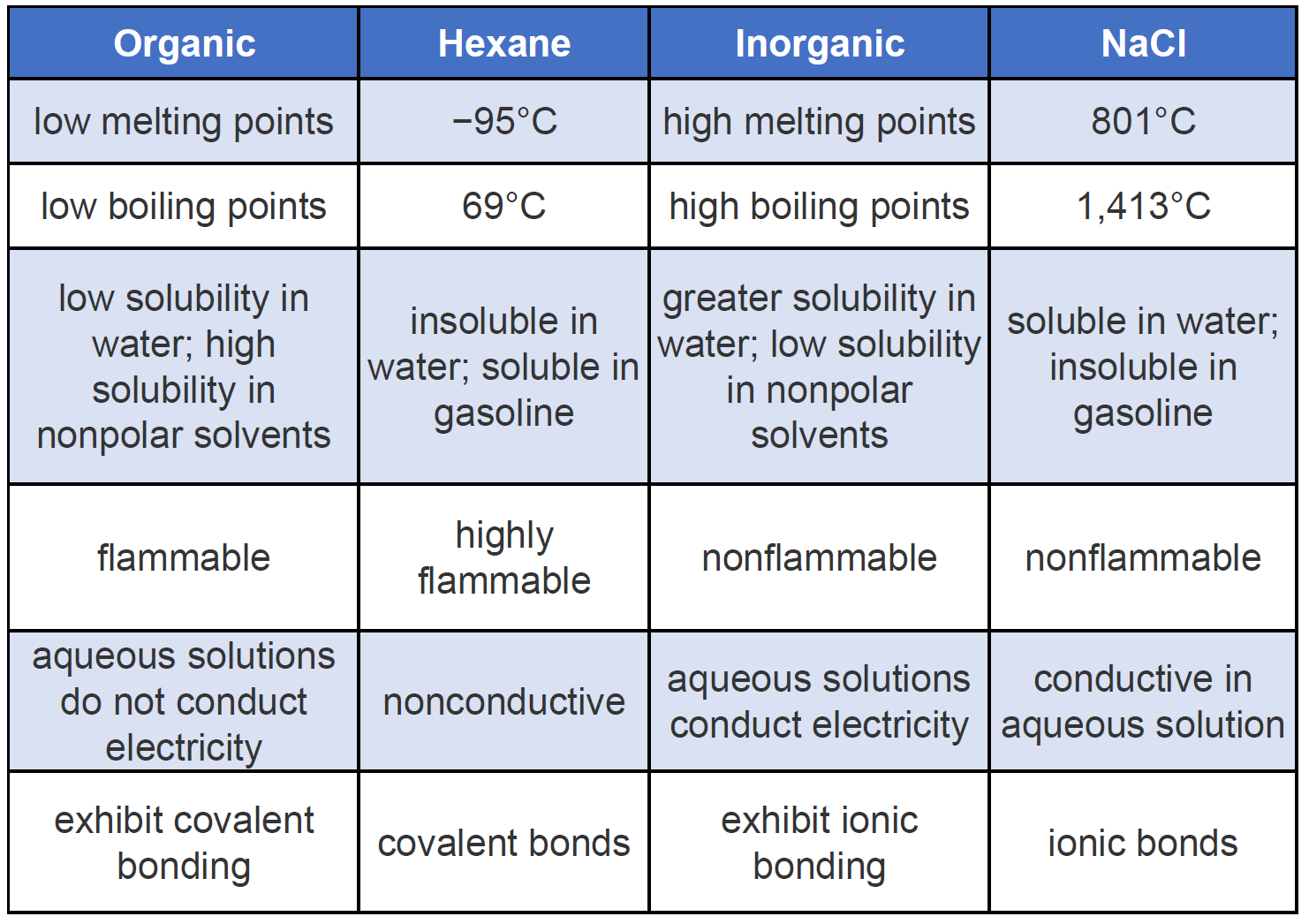
Ch105 Chapter 7 Alkanes And Halogenated Hydrocarbons Chemistry
Difference Between Organic And Inorganic Compounds

Examples Of Metals And Inorganic Compounds Used In Medicine Rat Oral Download Scientific Diagram

Inorganic Compounds An Overview Sciencedirect Topics

Difference Between Organic And Inorganic Compounds Definition Structure Properties Chemistry Lessons Chemistry Education Study Chemistry

List Of Inorganic Chemicals Used In The Experiments Download Table

Organic Or Inorganic Texas Gateway
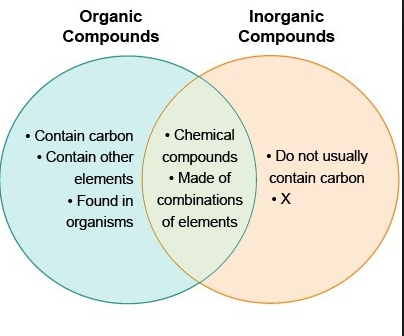
14 Difference Between Organic And Inorganic Compounds With Examples Viva Differences

Organic Or Inorganic Texas Gateway
Inorganic Compound Chemical Compound Britannica

Inorganic Compounds Essential To Human Functioning Anatomy And Physiology I

Organic Or Inorganic Texas Gateway

Inorganic Compounds As Pigments In Paints Infographic Chemistry Com Pk